Experiment 2, Cd test: percentages of K HNO3 of fragments of the alga... | Download Scientific Diagram

Selective adsorption of Pd(II) over Ag(I) in nitric acid solutions using nitrogen-donor-type adsorbents - ScienceDirect

Salicylic Acid Nitration by Means of Nitric Acid/Acetic Acid System: Chemical and Kinetic Characterization | Organic Process Research & Development

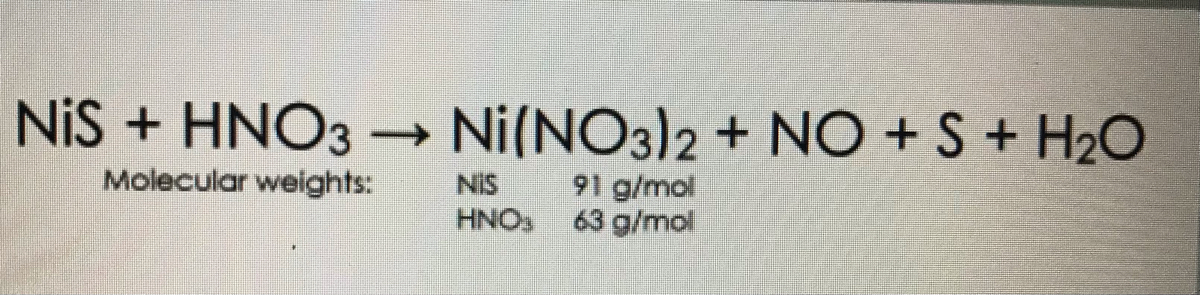
How to balance the following chemical equation S+HNO3---- H2SO4 + NO2+H2O by ABCD Method - Science - Chemical Reactions and Equations - 16660679 | Meritnation.com
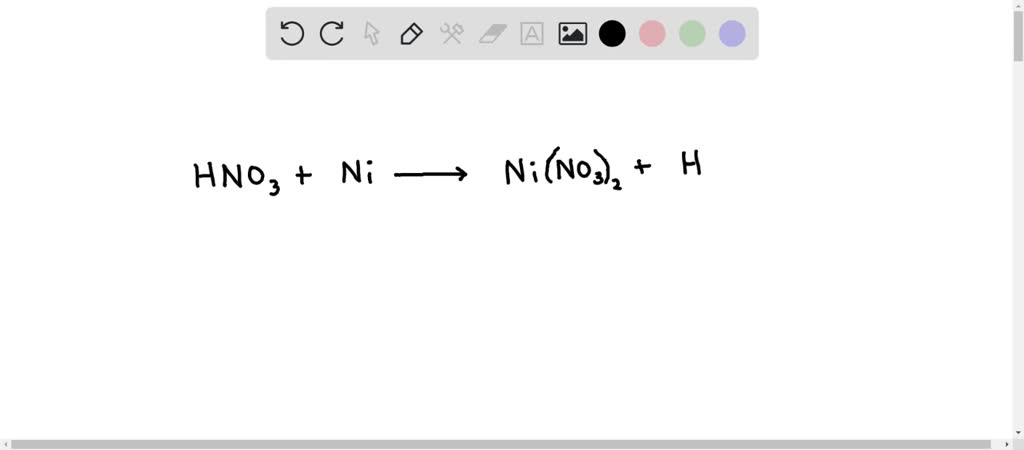
SOLVED: Write the balanced equation for the reaction of nitric acid and nickel metal reacting to form nickel(II) nitrate and hydrogen gas. Don't worry about states or making the subscripts subscript. Write
![PDF] Studies on Ni-Sn intermetallic compound and P-rich Ni layer at the electroless nickel UBM-solder interface and their effects on flip chip solder joint reliability | Semantic Scholar PDF] Studies on Ni-Sn intermetallic compound and P-rich Ni layer at the electroless nickel UBM-solder interface and their effects on flip chip solder joint reliability | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/dd3b9c617b9af5ce8d4299a0c71bb33d2e456dc1/2-Table1-1.png)
PDF] Studies on Ni-Sn intermetallic compound and P-rich Ni layer at the electroless nickel UBM-solder interface and their effects on flip chip solder joint reliability | Semantic Scholar

How many grams of concentrated nitric acid solution should be used to prepare 250 mL of 2.0 M HNO3? - India Site