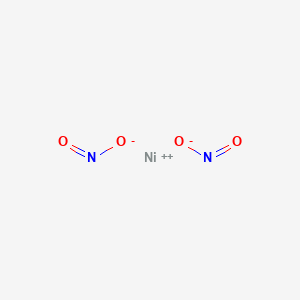
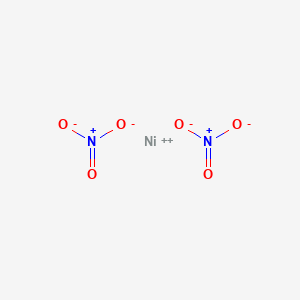
Which of the following compounds exhibit geometric isomers? a. Pt(NH3)2Cl2 (square planar) b. Co(H2O)2Cl3 c. Ni(NH3)4(NO2)2 d. K2CoCl4 | Homework.Study.com

Spectra of the solid complexes spread on filter paper, at 25 °C: [Ni II... | Download Scientific Diagram
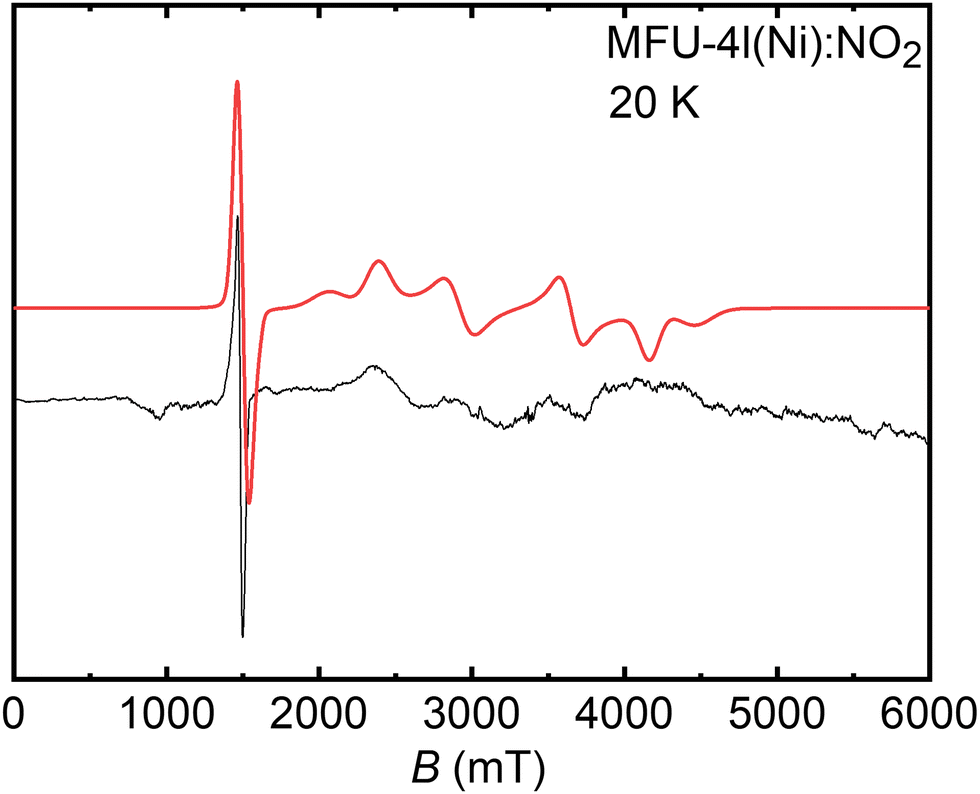
Unveiling the atomistic and electronic structure of Ni II –NO adduct in a MOF-based catalyst by EPR spectroscopy and quantum chemical modelling - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D3CP01449E
![laup (d) dsp2 (c) spºd 18. The correct order the wavelength of absorption the given complexes in the visible region is (a) [Ni(NO2)614- < [Ni(NH3)]2+ < [Ni(H20)6]2+ (b) [Ni(NO2)614- < [Ni(H20)]2+ < [ laup (d) dsp2 (c) spºd 18. The correct order the wavelength of absorption the given complexes in the visible region is (a) [Ni(NO2)614- < [Ni(NH3)]2+ < [Ni(H20)6]2+ (b) [Ni(NO2)614- < [Ni(H20)]2+ < [](https://toppr-doubts-media.s3.amazonaws.com/images/6111003/19c14228-dbc4-4bea-9c15-ad37a9f155d8.jpg)
laup (d) dsp2 (c) spºd 18. The correct order the wavelength of absorption the given complexes in the visible region is (a) [Ni(NO2)614- < [Ni(NH3)]2+ < [Ni(H20)6]2+ (b) [Ni(NO2)614- < [Ni(H20)]2+ < [

Redox Chemistry of Nickel(II) Complexes Supported by a Series of Noninnocent β-Diketiminate Ligands | Inorganic Chemistry
![Catalytic activity of K2Ba[Ni(NO2)6] on the thermolysis and laser ignition of CL-20, FOX-7 and TKX-50 - ScienceDirect Catalytic activity of K2Ba[Ni(NO2)6] on the thermolysis and laser ignition of CL-20, FOX-7 and TKX-50 - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022369721004777-gr2.jpg)
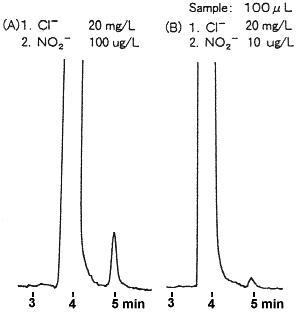
Catalytic activity of K2Ba[Ni(NO2)6] on the thermolysis and laser ignition of CL-20, FOX-7 and TKX-50 - ScienceDirect

The once-elusive Ni(IV) species is now a potent candidate for challenging organic transformations - ScienceDirect


![Schematic structure of the [Ni(Et 4 dien)(η 2 -O,ON)(η 1 -NO 2 )]... | Download Scientific Diagram Schematic structure of the [Ni(Et 4 dien)(η 2 -O,ON)(η 1 -NO 2 )]... | Download Scientific Diagram](https://www.researchgate.net/publication/265137173/figure/fig1/AS:392353174048768@1470555694916/Schematic-structure-of-the-NiEt-4-diene-2-O-ONe-1-NO-2-system-The-four.png)