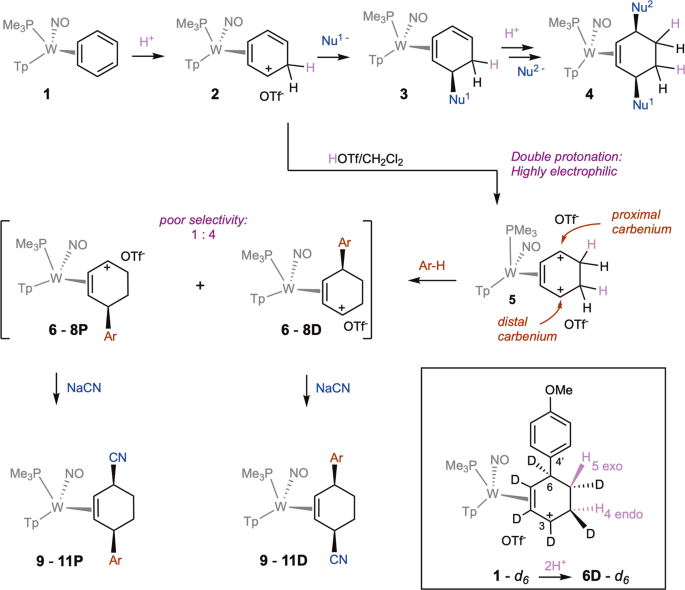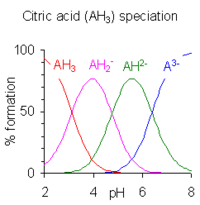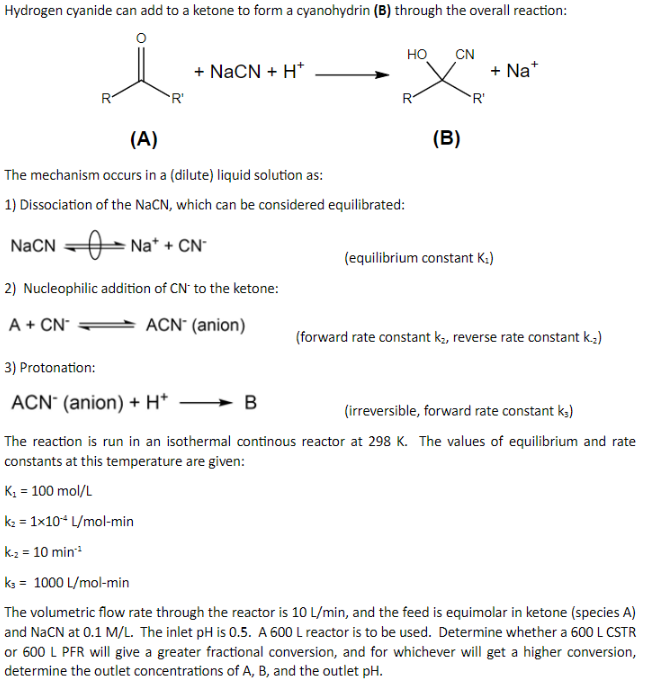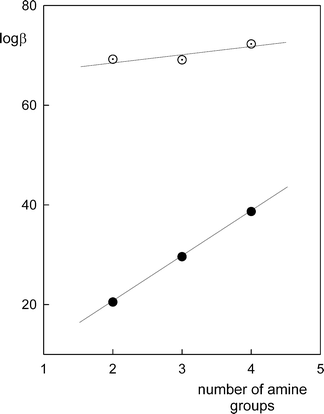
Equilibrium study on the interaction of phytic acid with polyamines and metal ions - Metallomics (RSC Publishing) DOI:10.1039/C1MT00031D

Reliability of protonation constants of vildagliptin dimers by the regression analysis of pH-titration data – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka
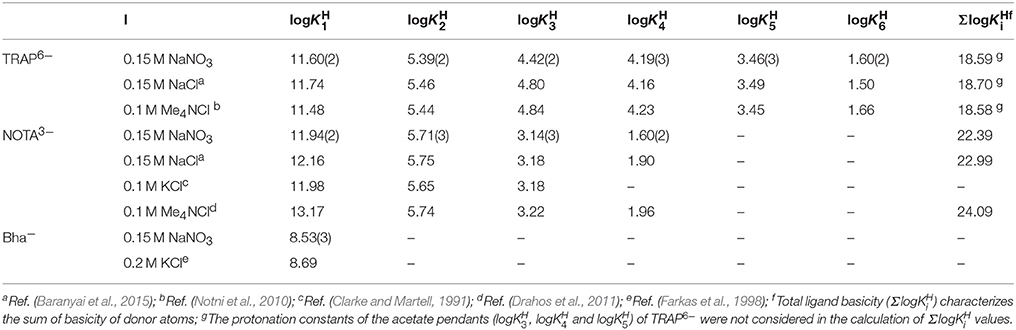
Frontiers | Equilibrium Thermodynamics, Formation, and Dissociation Kinetics of Trivalent Iron and Gallium Complexes of Triazacyclononane-Triphosphinate (TRAP) Chelators: Unraveling the Foundations of Highly Selective Ga-68 Labeling

Full article: Stability constants and complex formation equilibria between iron, calcium, and zinc metal ions with vitamin B9 and glycine

Protonation Constants and Thermodynamic Properties of Amino Acid Salts for CO2 Capture at High Temperatures | Industrial & Engineering Chemistry Research

PDF) Spectrophotometric Determination of Protonation Constants of L-Dopa in Dimethylformamide-Water Mixtures | GRD JOURNALS - Academia.edu

CPT Protonation Constants log β pqs and the Overall Formation Constants... | Download Scientific Diagram

Effect of Dielectric constant on Protonation Equilibria of Glycylglycine in Aqueous Solutions of propylene glycol and Dioxan | Semantic Scholar

Protonation Constants and Thermodynamic Properties of Amino Acid Salts for CO2 Capture at High Temperatures | Industrial & Engineering Chemistry Research

Solution Structure and Acid‐Base Properties of Reduced α‐Conotoxin MI - Faragó - 2021 - Chemistry & Biodiversity - Wiley Online Library

Communication. Determination of concentrations and protonation constants of monofunctional acids and bases by difference potentiometry - Analyst (RSC Publishing)
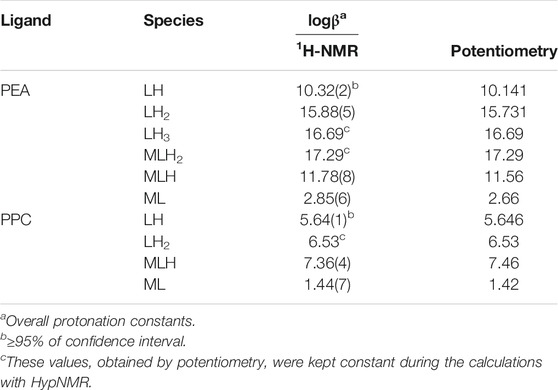
Frontiers | Speciation Study on O-Phosphorylethanolamine and O-Phosphorylcholine: Acid–Base Behavior and Mg2+ Interaction

Protonation constants and thermodynamic properties of amines for post combustion capture of CO2 | Semantic Scholar
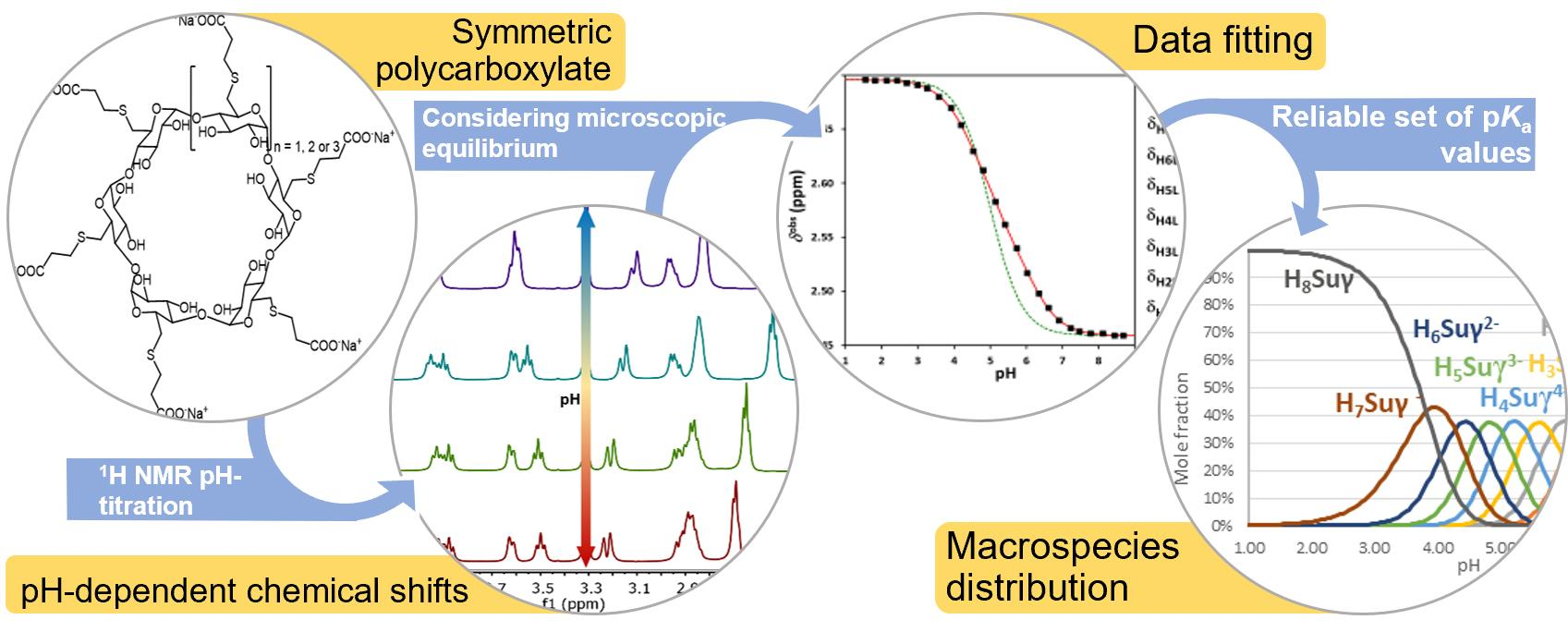
IJMS | Free Full-Text | Fully Symmetric Cyclodextrin Polycarboxylates: How to Determine Reliable Protonation Constants from NMR Titration Data
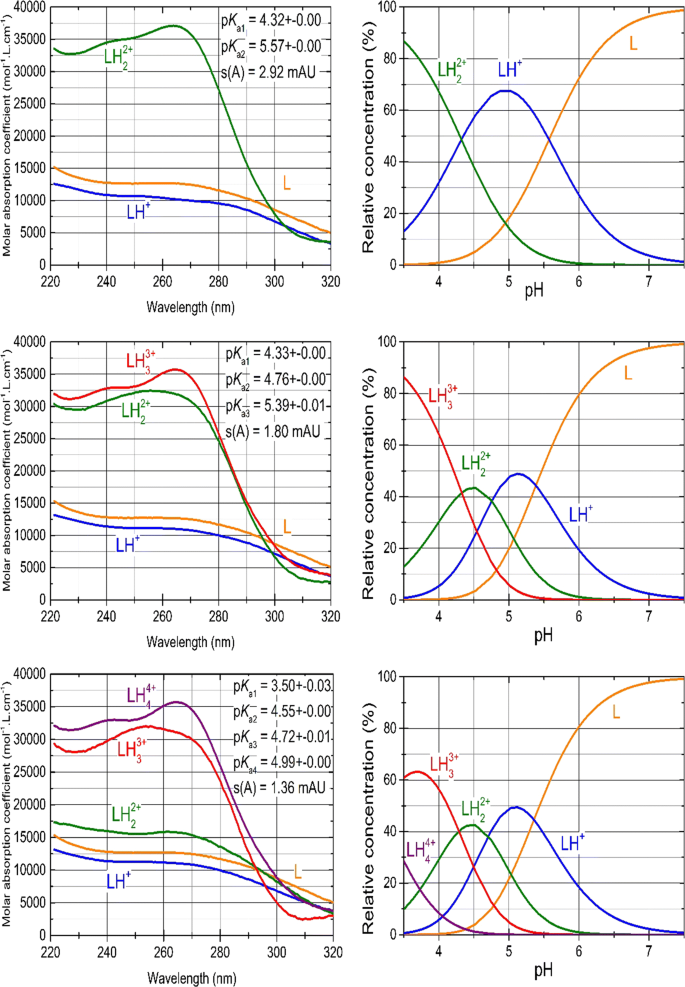
A Search for the Protonation Model with Thermodynamic Dissociation Constants and (Extra)-Thermodynamics of Nilotinib Hydrochloride (TASIGNA) | Journal of Solution Chemistry